QUALITY ASSURANCE
QUALITY ACCREDITATION
RIA Laboratory Co.,Ltd. or "RIA Lab" pays attention to the Quality Control System in every step of the analysis process. We operate based on the benefit of the client as the first under the concept "Specimens is Life", therefore, every process is controlled to get accurate and quality analysis results.
- Receiving specimens from clients Delivery of specimens Temperature is controlled and delivered to the laboratory under appropriate conditions for each analysis such as clinical chemistry, hematology, immunology, microbiology, etc.
- Inspection of specimens Specimen preparation before analysis With Check/Count Specimens are correct with the delivery note/Request for analysis service every time.
- Analysis process That has introduced advanced technology Used in the analysis process as follows
- LABORATORY INFORMATION SYSTEM (LIS) is used to connect the analysis results from the analyzer to the reporting system.
- VITROS® Automation Solutions (VAS) is a fully automated system that controls and manages the inspection process of the analyzer so that it can be executed quickly. Since the process Pre-analysis, Analysis and Post-analysis
- Automation Machines use sophisticated analysis tools and provide accurate and highly accurate diagnosis results.
- VITROS® 5,1 / FS Chemistry System Clinical Chemistry Analyzer with Dry Chemistry Technology
- VITROS® 5600 Integrated System Clinical Chemistry and Immunology Analyzer
- VITROS® 3600 Immunodiagnostic System Immunological Analyzers
- ORTHO AutoVue® Innova System, Blood Bank Analyzer
- Sysmex® Automation System Hematology Analyzers
- Biorad® D-100 HbA1c Hemoglobin Level Analyzers (HPLC)
- Biorad® Variant II Hb Typing by HPLC
- BD BACTECTM Blood Culture Blood culture media
In addition, RIA Laboratory Co.,Ltd. also uses other medical analysis products and reagents. With quality and providing accurate and accurate test results.
4. Process after analysis The online result reporting model has been used to provide analysis results in order to increase the speed in receiving results.
QUALITY CONTROL
Internal Quality Control System (Internal Quality Control: IQC)
Examining the laboratory conditions in each step To ensure that it is within the specified criteria (Medical Technology Council, Bangkok Post.) And there is a quality audit (Quality Audit) periodically that the laboratory has fully complied with the quality system And upheld by employees at all levels (G-08 TISI). This must cover the examination of the management of the quality system Technical, technical, and safety systems (ETA)
Laboratory Quality Assessment by External Quality Assessment (EQA)
External quality assessment (EQA) is an important way to assure laboratory quality assurance. With the objective of having an agency to inspect the quality of the test results of the laboratory It can be seen that the laboratory standards must be evaluated regularly by external organizations. That is, must participate in the External Quality Assessment Scheme with any organization. To support the quality system of that laboratory However, the evaluation of the project has a great impact on the laboratory. The laboratory therefore must have a good understanding of the assessment. Both to consider the project to join as a member That it is a good quality project or not And most importantly The information obtained from the project is able to properly utilize the quality system of the laboratory.
RIA Laboratory has passed the laboratory Quality Certification as follows : 3 Standards
- ISO 15189 - from the Bureau of Laboratory Standards Department of Medical Sciences Ministry of Public Health
- ISO 15190 - Safety in medical laboratories Bureau of Laboratory Standards Department of Medical Sciences Ministry of Public Health.


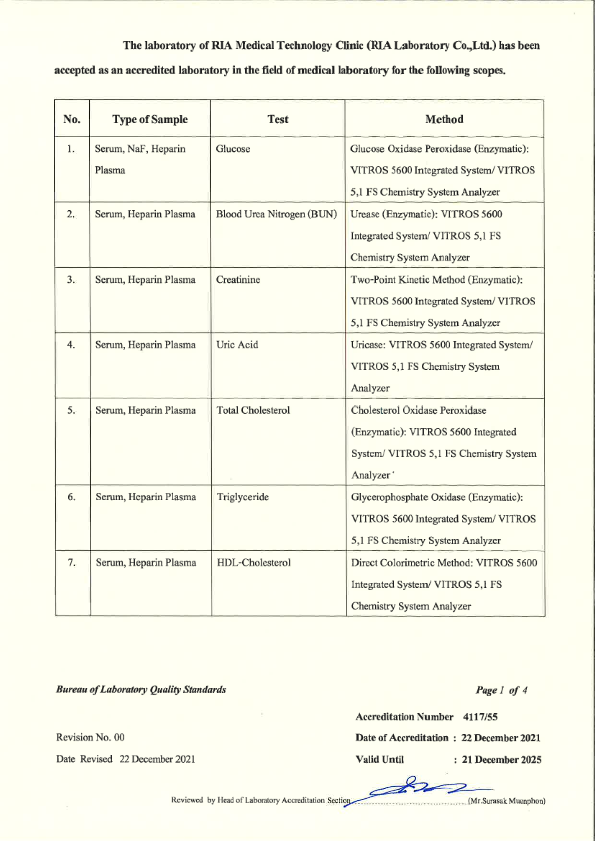
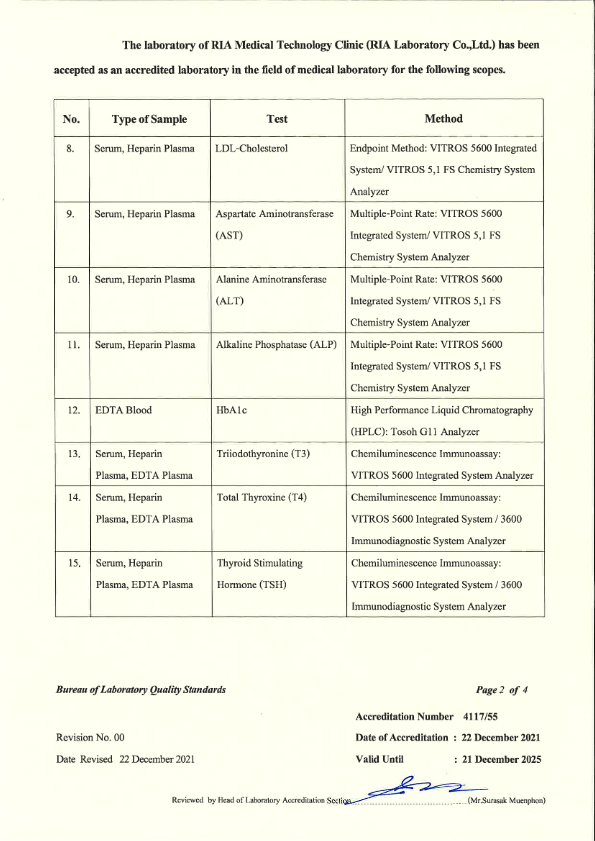
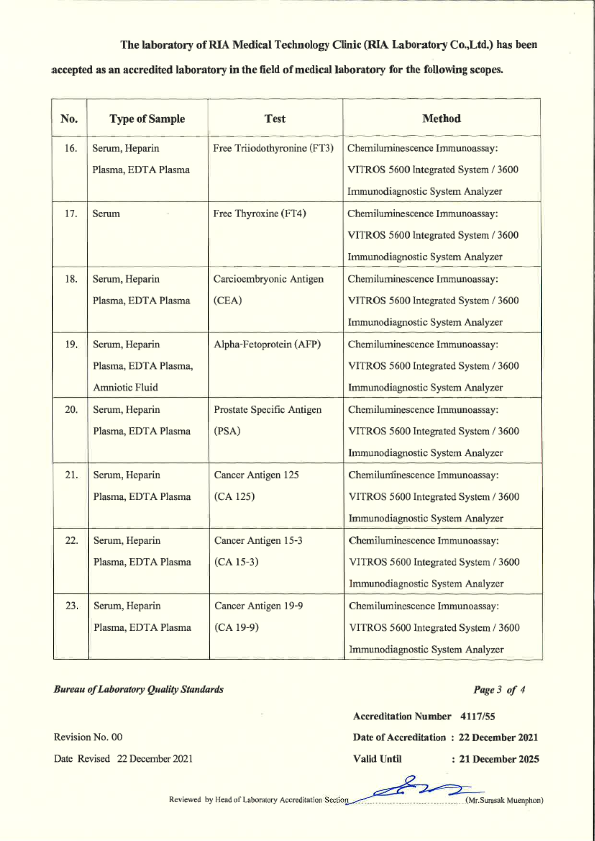
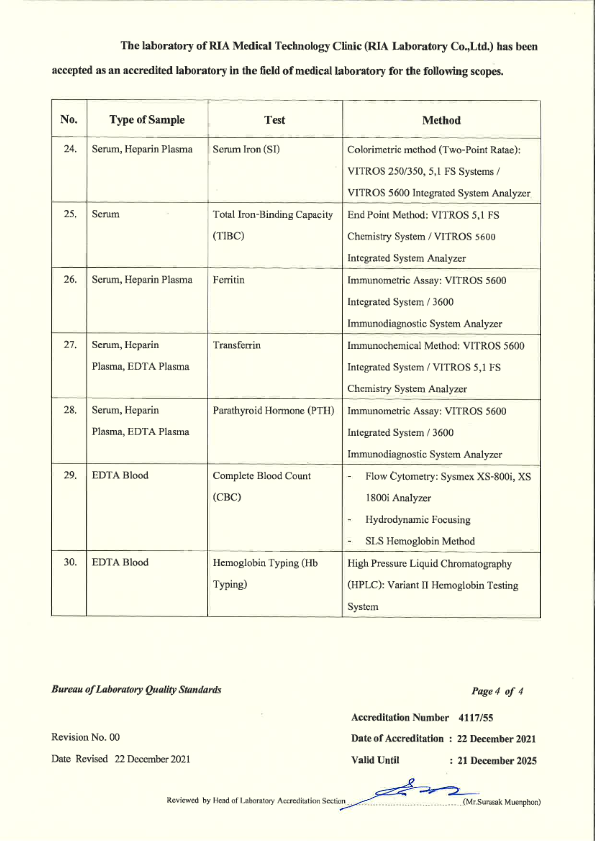
- Laboratory Accreditation - Quality Management System Accreditation for Medical Technology Standards For medical laboratories By the Medical Technology Council

This website uses cookies. to increase work efficiency if you accept Indicates that you have given your consent to the cookies of the website. and our privacy policy.


